The medical courier market reached $9.3 billion in 2022 and is projected to hit $16.2 billion by 2032, growing at a 6.42% CAGR (Medical Courier Services Market Report, 2023). The courier companies winning those contracts aren't the cheapest; they're the ones who understand what healthcare procurement teams evaluate.
That evaluation comes down to operational evidence. Ethic Transport, a Belgium-based courier company, secured a long-term partnership with GLS within six months of founding because they could demonstrate a 100% parcel delivery rate from day one. Not a promise of reliability. Proof of it.
We've included a nine-section bid proposal template built for medical courier operations. Four of those sections are medical-specific: chain of custody, temperature monitoring, incident response, and compliance certifications. Most competing bid guides skip all four. Procurement teams score you on those sections when choosing between courier bids. Adapt the template for your next submission.
If you're looking for individual delivery gigs rather than business contracts, platforms like GoShare list opportunities. The strategies here are for .
What you need before you bid
Medical courier contracts require specific vehicles, insurance, certifications, and equipment before healthcare facilities will consider your bid. Healthcare facilities carry their own regulatory liability when they outsource specimen transport. A HIPAA violation by your driver exposes them, not just you. Procurement teams check compliance first because it's their risk filter. Miss any one requirement, and your bid is disqualified before they even look at delivery success rates or technology.
Vehicle requirements
Medical specimen transport demands more than a clean van. Equip vehicles with temperature-controlled storage (both refrigerated and ambient compartments), GPS tracking systems, secure lockable compartments for pharmaceuticals, and dedicated biohazard transport areas separated from the driver cabin.
Vehicle classes range from sedans for document and small specimen runs to cargo vans and box trucks for equipment and bulk pharmaceutical deliveries. Non-CDL drivers can handle most medical courier routes - a strong selling point for smaller clinics and pharmacies that need affordable, reliable service without CDL-associated costs.
Insurance coverage
Medical courier operations require four insurance types to cover their liability profile: general liability, commercial auto, cargo, and inland marine. Each addresses a different risk category - from third-party injury claims to high-value medical materials in transit.
- General liability protects against third-party bodily injury and property damage claims during pickups and deliveries.
- Commercial auto insurance covers vehicles used for business purposes - personal auto policies exclude commercial medical transport.
- Cargo insurance covers the value of medical materials in transit, including specimens, pharmaceuticals, and equipment.
- Inland marine insurance covers goods in transit over land that standard cargo policies may exclude - particularly high-value medical equipment and temperature-sensitive biologics.
Procurement teams verify all four coverage types during the bid evaluation process. Missing any one can disqualify your proposal before evaluators review your operational capabilities.
Certifications and training
Medical courier companies need four core certifications before healthcare facilities will consider their bids: HIPAA (Health Insurance Portability and Accountability Act) training, OSHA Bloodborne Pathogens certification, HAZMAT endorsement, and medical specimen handling credentials. These four prove your team meets the regulatory bar for medical transport.
- HIPAA training is required for anyone handling protected health information, including patient specimens and medical records.
- OSHA Bloodborne Pathogens (BBP) certification is mandatory for employees handling biological specimens. It covers exposure control, PPE requirements, and incident response.
- HAZMAT endorsement is required for transporting certain pharmaceutical and chemical materials classified under DOT (Department of Transportation) regulations.
- Medical specimen handling certification - programs through organizations like Integrity Delivers - covers chain of custody, temperature management, and regulatory compliance.
Meeting these requirements before approaching healthcare facilities positions your company as a credible bidder from the first conversation.
Equipment checklist
Every vehicle in your fleet needs standard medical transport equipment before you can bid on healthcare contracts. Stock every vehicle with insulated coolers and dry ice for cold-chain specimens, continuous temperature monitors with digital alert systems, PPE (gloves, goggles, gowns), biohazard spill kits and cleanup supplies, tamper-evident packaging, and chain of custody documentation forms.
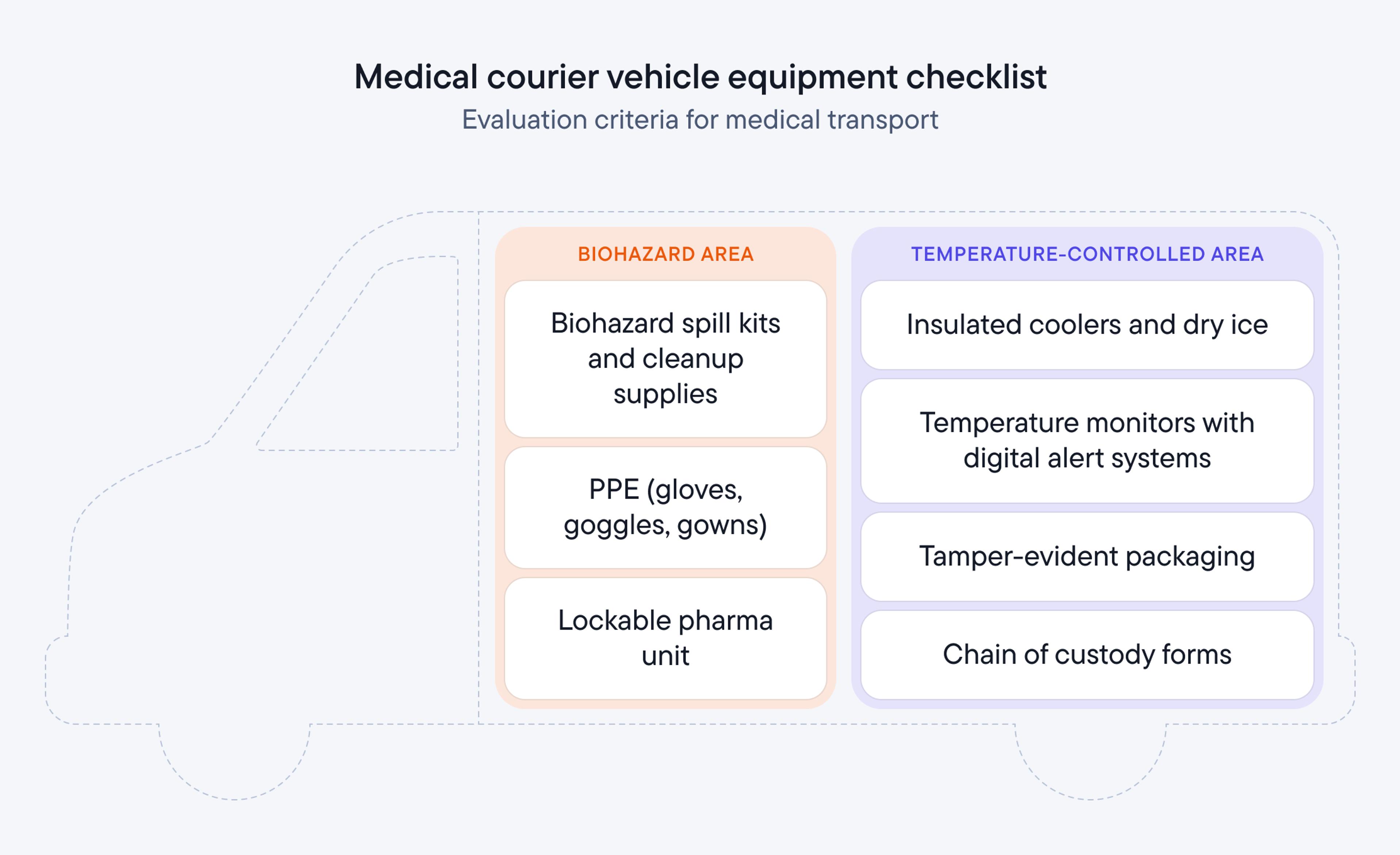
Audit every vehicle against this list before submitting your first bid - procurement teams check equipment readiness early in the evaluation process.
HIPAA, OSHA, and DOT - What compliance looks like in practice
Medical courier compliance sits across three regulatory frameworks. HIPAA covers patient data and specimen privacy, while OSHA's Bloodborne Pathogens Standard governs safe handling of biological materials. DOT regulations control hazardous material transport. A compliance gap in any of the three puts the healthcare facility's own accreditation and patient safety record at risk, which is why procurement teams treat compliance documentation as a qualifying gate before evaluating anything else. HIPAA penalties range from $100 to $50,000 per violation, with annual maximums up to $1.5 million per violation category.
HIPAA requirements for medical couriers
HIPAA applies the moment your drivers handle anything tied to a patient - specimens, records, lab results, prescription deliveries. In a courier context, compliance means maintaining chain of custody documentation at every handoff, securing transport against unauthorized access, keeping current staff training records, and having breach notification procedures ready. Each requirement looks like this operationally:
- Chain of custody documentation at every handoff point (pickup, transport, delivery). Each transfer must be logged with timestamps, handler identification, and specimen condition notes.
- Secure transport protocols that prevent unauthorized access to patient-linked materials during transit. Lockable compartments, sealed packaging, and tamper-evident containers are the minimum.
- Staff training records proving every driver completed HIPAA training within the last 12 months. Procurement teams request these during bid evaluation.
- Breach notification procedures documenting who to contact and within what timeframe if a privacy incident occurs during transport.
OSHA Bloodborne Pathogens Standard
The OSHA BBP standard applies to every courier handling biological specimens - blood samples, tissue samples, cultures, and any material that could contain bloodborne pathogens. Compliance requires a written exposure control plan, appropriate PPE for all specimen handlers, documented incident reporting procedures, and annual refresher training. Healthcare facilities verify BBP compliance during bid evaluation and may request your exposure control plan as part of the submission.
- The exposure control plan documents which job classifications involve exposure risk, the control methods in place (engineering controls, work practice controls, PPE), and the procedures for post-exposure evaluation.
- PPE requirements include gloves for all specimen handling, plus goggles and gowns when handling open or potentially compromised containers. Employers must provide PPE at no cost to drivers.
- Any exposure event (needle stick, container breach, spill) must be documented and reported immediately. Post-exposure medical evaluation must be offered within 24 hours.
- Refresher training is required every 12 months, covering updated procedures, new hazards, and incident review.
Maintaining current BBP documentation protects your drivers and demonstrates to procurement teams that your operation takes biological specimen handling seriously.
DOT regulations
DOT regulations govern how medical materials classified as hazardous are packaged, labeled, and transported. Infectious substances, certain pharmaceuticals, and chemical reagents all fall under DOT classification requirements. Non-compliance carries fines and can result in contract termination, so courier companies need to know which materials in their typical runs require DOT-specific handling, packaging, and driver training.
- Infectious substances fall under DOT Class 6.2. Certain pharmaceuticals and chemicals may fall under additional classifications. Identify which materials in your typical runs carry DOT classification requirements before bidding.
- UN-specification packaging is required for Category A infectious substances. Category B substances (most routine medical courier materials) require less stringent but still specific triple-packaging with absorbent material.
- All drivers need general awareness training. Those handling hazmat-classified materials need function-specific training and security awareness training. Refresher training is required every three years.
Ensuring every driver understands which materials in their routes carry DOT requirements prevents violations that could jeopardize existing contracts and disqualify future bids.
Every driver and handler on your team needs training across HIPAA, OSHA BBP, and DOT before you submit your first bid. Procurement teams verify training records during evaluation - expired certifications are an automatic disqualification. Training resources like Integrity Delivers training programs offer live webinars, online courses, and certification programs covering all three frameworks. HIPAA and BBP require annual renewal. DOT refresher training runs on a three-year cycle. Build renewal dates into your compliance calendar so certifications don't lapse between contracts.
Where to find medical courier contracts
Medical courier contracts come through three main channels: government procurement portals, direct outreach to healthcare facility procurement departments, and industry associations. Government contracts are public and searchable, while private-sector contracts require relationship building and RFP monitoring.
Government procurement portals
Government healthcare facilities - VA hospitals, county health departments, state laboratories - post courier contracts on public procurement platforms. These contracts are searchable, publicly available, and often recurring, making them a reliable pipeline source for medical courier companies. Register on the major platforms and set up keyword alerts to receive notifications when new courier service solicitations are posted in your service area.
- SAM.gov is the federal System for Award Management. Search for active solicitations under NAICS codes related to courier and express delivery services. Register your business to receive notifications.
- BidNet Direct aggregates government RFPs, including laboratory courier services across state and local agencies.
- State and county health department procurement portals are another source - many states post courier service RFPs directly on their health department websites. Check procurement pages for your target service area.
Getting in front of hospital procurement
Private-sector medical courier contracts come through direct relationships with hospital and university procurement departments. Start by searching the facility's website for their procurement or purchasing department page - most hospital systems publish vendor registration forms and contact details there. Call the procurement office directly and ask for the vendor onboarding process. Don't contact clinical staff - they don't handle courier contracts.
Show the procurement contact your compliance documentation, delivery success rates, and technology setup. Large health systems maintain approved vendor lists, and getting on that list positions your company for future RFP invitations even if there's no immediate contract. Ask the procurement contact specifically what their vendor qualification process requires - some systems need a capabilities presentation, others want a written submission.
Industry associations and networking
Industry associations connect medical courier companies with contract opportunities, procurement contacts, and regulatory updates that don't appear on public portals. Membership in the right organizations signals professionalism to healthcare procurement teams and gives you early access to RFPs circulated through member networks.
- The Customized Logistics and Delivery Association (CLDA) is the primary trade association for last-mile delivery companies. Membership provides access to contract opportunities and industry networking.
- HIMSS conferences are healthcare IT events where courier companies can connect with facility procurement and logistics teams.
- Medical Courier Connection offers industry-specific networking and resources for medical courier operators.
- The World Health Organization (WHO) issues international RFPs for courier services across global health programs.
Set up alerts on government procurement platforms and check industry association job boards weekly.
Beyond healthcare facilities
Healthcare facility procurement is one pipeline, but not the only one. Pharmaceutical companies, research universities, biotech labs, blood banks and tissue banks, dental labs, veterinary clinics, and insurance companies also contract courier services for specimen and material transport.
These organizations post RFPs through different channels - pharmaceutical companies often use dedicated procurement platforms, while smaller labs may contract through direct outreach. If your operation handles temperature-sensitive biologics, these verticals are worth pursuing alongside healthcare facility contracts.
What healthcare procurement teams evaluate
Healthcare procurement teams evaluate medical courier bids on seven primary criteria: documented delivery success rates, compliance certification evidence, temperature monitoring protocols, chain of custody documentation, technology capabilities, response time and SLA guarantees, and references from similar healthcare facilities. The courier companies that present operational evidence - not promises - win contracts.
Most bid advice focuses on what to include in your proposal, but ignores what the buyer on the other side of the table prioritizes when scoring it.
What procurement teams score you on
Each criterion below shows what procurement teams look for and how to provide the evidence they need.
Criterion | What procurement looks for | How to provide evidence |
|---|---|---|
Delivery success rate | Documented track record with specific percentages, not vague reliability claims | Monthly delivery reports showing success rates: Ethic Transport, a Spoke Dispatch customer, demonstrated a 100% parcel delivery rate - the kind of documented proof that procurement teams evaluate |
Compliance documentation | Current HIPAA training records, OSHA BBP certifications, DOT compliance for all drivers | Organized compliance folder with expiration dates, training completion certificates, and renewal schedules |
Temperature monitoring | Continuous temperature logs from previous medical deliveries, not just equipment specifications | Digital temperature monitoring data exports showing unbroken cold-chain records across deliveries |
Chain of custody | Documented handoff protocols with timestamps, handler IDs, and condition notes at every transfer point | Sample chain of custody logs from previous healthcare client deliveries |
Technology capabilities | Real-time GPS tracking, electronic proof of delivery, automated recipient notifications, route optimization, client visibility portals | Live demo or screenshots of courier dispatch software showing tracking, POD records, and notification logs |
Response time and SLA guarantees | Defined time windows for different delivery types (stat, routine, scheduled) with penalty frameworks for missed windows | Documented SLA performance history showing time window adherence rates across previous healthcare contracts |
References | Named healthcare facility references who can speak to your service quality and reliability | Contact list of current healthcare clients willing to serve as references, with contract duration noted |
Client portals that give facilities direct delivery visibility score particularly well. Spoke Connect, for example, gives healthcare facilities a direct portal to track shipments, upload delivery requirements, and access proof of delivery records - eliminating the phone calls and email chains that slow down courier-facility communication.
As the founder of Ethic Transport put it:
They knew that if we took 100 parcels, we'd deliver all 100 parcels.
That level of documented reliability is what separates winning bids from generic proposals.
How to prove it, not promise it
Structure your bid evidence around four principles:
- Quantify everything. "99.2% delivery success rate across 14,000 deliveries in 2025" beats "excellent track record."
- Show, don't claim. Attach temperature monitoring logs, chain of custody samples, and delivery performance reports as appendices.
- Match their scale. If bidding for a hospital system doing 200+ specimen pickups per day, reference experience at comparable volume - not your performance on five-delivery runs.
- Address failure handling. Procurement teams know failures happen. Show your incident response protocol, not just your success rate.
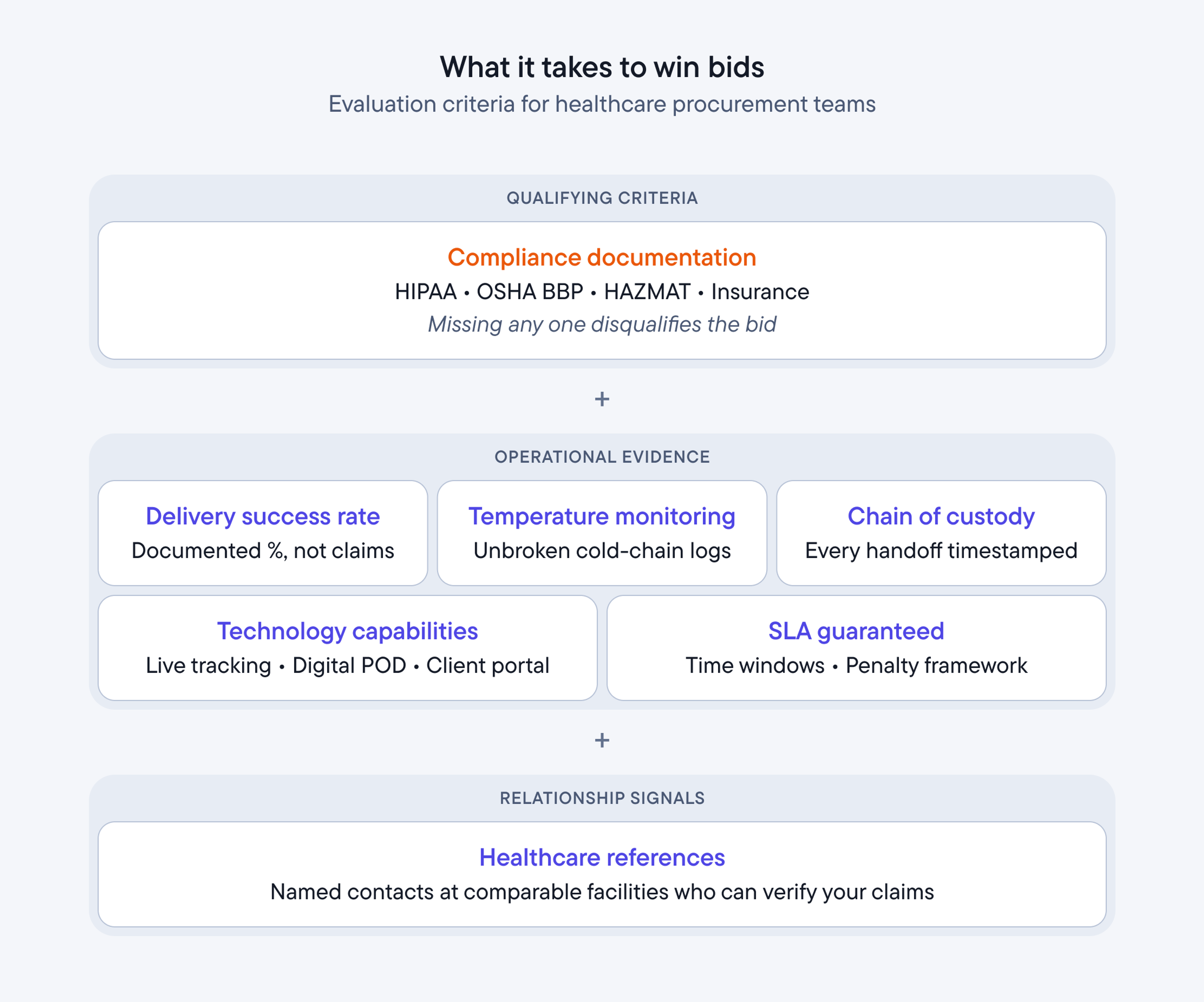
The courier companies that win contracts are the ones who make it easy for procurement teams to verify every claim in the bid.
Medical courier bid proposal template
As we mentioned, a winning medical courier bid proposal includes nine sections: five standard business sections and four medical-specific sections that separate your bid from generic courier submissions. The four medical sections - chain of custody, temperature monitoring, incident response, and compliance certifications - are what procurement teams look for when evaluating medical courier bids specifically.
Most competing guides offer a generic bid outline with three to five sections. The most detailed competitor template covers eight sections - none specific to medical courier operations. The four medical-specific sections that follow address the exact criteria healthcare procurement teams evaluate.
Starting with operational technology that generates documented delivery metrics - rather than adding it later - gives them the evidence procurement teams needed to see from day one.
Bid Proposal Template
Free to use - no signup required
Submitted by: [Your company name]
Date: [DD/MM/YYYY]
Prepared for: [Client company name]
1. Company overview
[Provide a brief description of your company, years in operation, geographic coverage, and any relevant certifications or accreditations.]
2. Scope of services
We propose to provide the following delivery services:
- Same-day and next-day parcel delivery
- Scheduled multi-drop routes
- Returns management and reverse logistics
- [Add/remove services as applicable]
3. Pricing structure
Service | Unit | Rate |
|---|---|---|
Standard delivery (0-5kg) | per parcel | [X.XX] |
Heavy freight (5-30kg) | per parcel | [X.XX] |
Fuel surcharge | % of base rate | [X] % |
4. Technology and tracking
[Describe your dispatch software, real-time tracking capability, proof of delivery process, and any API or integration options relevant to the client.]
5. SLAs and performance guarantees
- On-time delivery rate: [X]% or above
- Claims resolution: within [X] working days
- Dedicated account manager: [Yes/No]
- Escalation procedures: [Describe]
- Reporting cadence: [Weekly/Monthly/Per-delivery]
6. Chain of custody documentation
- Handoff procedure: [Describe pickup, transport, and delivery logging process]
- Custody log format: [Digital/Paper - specify system]
- Tamper-evident packaging: [Describe standards]
- Audit trail: [Describe how unbroken chain of custody is maintained and verified]
7. Temperature monitoring and compliance
- Cold-chain equipment: [List insulated containers, active cooling units, dry ice protocols]
- Monitoring process: [Continuous/Periodic - specify system and alert thresholds]
- Excursion protocol: [Describe response when temperature deviates from acceptable range]
- Calibration records: [Describe equipment calibration schedule and documentation]
8. Incident response protocol
- Spill containment: [Describe biological material breach procedures]
- Exposure response: [Immediate actions, medical evaluation timeline, reporting chain]
- Damaged specimen handling: [Describe procedures]
- Notification chain: [Who gets contacted, in what order, within what timeframe]
- Post-incident review: [Describe documentation and review process]
9. Compliance certifications
- HIPAA training records: [Completion dates for all drivers]
- OSHA BBP certification: [Current/Expiry date]
- HAZMAT endorsement: [If applicable - current/expiry date]
- DOT compliance documentation: [Describe]
- Insurance certificates: [General liability, commercial auto, cargo, inland marine - attach]
- Business licenses: [Current/Expiry date]
Ethic Transport's founder describes what enabled their operation to win a long-term GLS partnership within six months of founding:
The fact that we had Spoke Dispatch helped us operate successfully from the very beginning.
Below, we break down what to include in each section and why it matters to procurement teams.
1. Company overview
Introduce your courier company with specific relevance to medical transport. Include founding year, fleet size and vehicle types, service area (cities, regions, or states covered), years of medical courier experience, and key healthcare clients served (with permission). Point out any specializations - cold-chain logistics, specimen transport, pharmaceutical delivery - that match the facility's needs.
2. Scope of services
Detail the delivery types you handle: laboratory specimens, pharmaceuticals, medical equipment, confidential records. Specify coverage hours - standard business hours, extended hours, 24/7 availability, or on-call emergency service. Define your geographic range and any specialized capabilities such as hazmat transport, same-day delivery, or cold-chain logistics for temperature-sensitive biologics.
3. Pricing structure
Present per-delivery rates, per-mile rates, and volume-based pricing tiers. Include a surcharge schedule covering urgent requests, after-hours service, hazmat handling, and extended-distance deliveries. Offer contract term options - monthly, quarterly, annual - with volume discounts for longer commitments. Transparent pricing builds confidence during procurement review.
4. Technology and tracking capabilities
Describe your dispatch software capabilities: real-time GPS tracking for every active delivery, route optimization for time-sensitive specimens, digital proof of delivery features (signatures, photos, timestamps, geotags), automated recipient notifications via SMS or email, and analytics and performance reporting. Include system integration capabilities if the facility uses specific healthcare logistics platforms.
5. SLAs and performance guarantees
Your SLA section tells procurement teams exactly what performance levels you commit to and what happens when you fall short. Medical courier contracts typically benchmark at 98% or higher delivery success rates - anything lower signals inexperience with healthcare logistics. Specify delivery success rate targets, define time window guarantees for different delivery types (stat, routine, scheduled), and outline your escalation procedures when SLAs are at risk, any financial penalty framework for missed targets, and reporting cadence (weekly, monthly, or per-delivery performance dashboards).
6. Chain of custody documentation
Detail your specimen handling protocol from pickup to delivery. Document the handoff procedure at each transfer point - who signs, what gets logged, and how condition is recorded. Specify your custody log format (digital logs are preferred over paper for audit purposes). Describe tamper-evident packaging standards and how your digital audit trail provides an unbroken chain of custody record.
7. Temperature monitoring and compliance
Temperature excursions are the most common reason medical deliveries are rejected at the receiving facility, making this section essential to your bid's credibility. Document your cold-chain equipment specifications - insulated containers, active cooling units, dry ice protocols - and describe your continuous temperature monitoring process, including alert thresholds and escalation procedures when excursions occur. Include calibration records for monitoring equipment and storage-in-transit protocols for different specimen categories.
8. Incident response protocol
Procurement teams know that failures happen - what separates strong bids is a documented response plan that minimizes damage and maintains compliance. Your incident response section should cover spill containment procedures for biological material breaches, your biological exposure response plan (immediate actions, medical evaluation timeline, reporting chain), damaged specimen handling, and the notification chain: who gets contacted, in what order, within what timeframe. Include documentation requirements for every incident and your post-incident review process.
9. Compliance certifications
Prove your regulatory readiness with documentation, not claims. Compile all regulatory documentation in one organized package: HIPAA training records with completion dates for every driver, OSHA BBP certification, HAZMAT endorsements where applicable, DOT compliance documentation, insurance certificates (general liability, commercial auto, cargo, inland marine), and current business licenses. Organize by expiration date so procurement teams can verify currency at a glance.
Technology that wins contracts
Technology capabilities are now a standard procurement criterion for medical courier contracts. Healthcare facilities expect real-time GPS tracking, automated delivery notifications, electronic proof of delivery with timestamps and photos, and route optimization for time-sensitive specimens. The courier companies meeting the 98%+ delivery success threshold that procurement teams benchmark against are the ones using dispatch software to capture and prove that performance.
Technology is the mechanism for generating the operational evidence that procurement teams evaluate. The seven criteria from the evaluation framework - delivery success rates, compliance documentation, temperature monitoring, chain of custody, technology capabilities, response time guarantees, and references - all depend on last mile management software that captures and reports operational data.
Route optimization for time-sensitive deliveries
Medical specimens often have strict delivery windows - blood samples can become unusable within hours of collection, cultures require timely processing to remain viable, and pharmaceutical deliveries must arrive within temperature-safe timeframes. Route optimization using real-time traffic data, delivery time windows, and vehicle capacity constraints ensures your drivers hit those windows consistently. A last mile management platform handles this at fleet scale - optimizing across multiple drivers, routes, and delivery priorities simultaneously.
Spoke’s Google Maps partnership provides route optimization software that accounts for traffic patterns, delivery time constraints, driver preferences, and starting locations. For medical courier operations, this means planning routes around specimen pickup schedules and lab processing deadlines - not just the shortest distance.
Real-time tracking
Procurement teams increasingly require real-time delivery visibility as a contract condition. Fleet tracking lets dispatchers monitor every active delivery, intervene when delays develop, and provide accurate ETAs to healthcare facility contacts. The capability also generates the operational data - delivery times, route adherence, exception logs - that strengthens future contract bids.
Paul LeBlanc, owner of KW Delivery, describes the impact:
On the day we agreed to the FedEx contract, I was looking for apps to help my team manage the high-volume routes. Frankly, it would've been disastrous if I hadn't found Spoke Dispatch.
KW Delivery scaled from 50 to 500 deliveries per day. At that volume, a dispatcher can't track drivers by phone or memory - you need a dashboard showing every active delivery, so you can spot a delayed specimen run before it misses its processing window.
Electronic proof of delivery
Chain of custody documentation requires proof at every transfer point. Digital POD - signatures, photos, timestamps, notes, barcode scans, and geotags - replaces paper logs that procurement teams find unreliable. Photo geotags show the exact delivery location, resolving disputes and providing evidence for compliance audits. Proof of delivery for pickup points captures the full chain from specimen collection to lab delivery.
Recipient notifications and delivery confirmation
Automated delivery notifications reduce failed delivery attempts. When recipients know a medical delivery is approaching, they're prepared to receive it - increasing first-attempt success rates. In medical courier operations, a failed first attempt means specimens sit in transit longer, risking degradation before they reach the lab. The On-Demand Retailer, a Spoke customer in Philadelphia, maintains a 99% delivery success rate. Recipient notifications are a key factor in that consistency.
The competitive advantage of operational technology
The gap between tech-enabled courier companies and legacy operations is widening - and healthcare procurement teams notice. Courier companies that offer real-time tracking, instant proof of delivery, and client-facing visibility portals win contracts over competitors still relying on paper logs and next-day reporting.
As one RoadRunner representative put it:
"Lots of clients are still very old school in terms of their tech systems. No ETAs, no ability to track the drivers. POD that they can't access for hours or even until the next day! It's just old and clunky, so by using Spoke Dispatch, we definitely have the advantage."

That difference is visible in every procurement evaluation. Facilities want verifiable, real-time evidence of delivery performance - not next-day paper reports. Investing in medical courier technology before you bid gives procurement teams confidence in your operational capabilities.
See how route optimization and real-time tracking work in practice - try Spoke Dispatch free for 7 days.
Questions you're probably asking
A medical courier contract is a formal agreement between a healthcare facility and a courier company for the regular transport of medical materials - specimens, pharmaceuticals, equipment, and records. Contracts typically specify delivery frequency, service hours, SLA requirements, compliance standards, and pricing terms. Most contracts run six to 12 months with renewal options.
Medical courier companies need commercial vehicle registration and insurance, HIPAA and OSHA BBP certifications for all specimen handlers, and clean driving records across the team. Some contracts also require HAZMAT endorsements.
Medical couriers transport laboratory specimens (blood, urine, tissue samples), pharmaceuticals, medical equipment, confidential patient records, imaging materials, and surgical supplies. Each material type has specific handling requirements - specimens require temperature control and chain of custody documentation, while pharmaceuticals may require DOT-classified hazmat handling.
Medical couriers operate under stricter regulatory requirements than general couriers. HIPAA compliance, OSHA BBP protocols, temperature-controlled transport, and chain of custody documentation are mandatory for medical materials. Standard couriers face none of these requirements. The regulatory overhead is why medical courier contracts typically pay higher rates - and why procurement evaluation is more rigorous.
A winning medical courier bid proposal needs nine sections - five standard business sections and four medical-specific additions, covering chain of custody, temperature monitoring, incident response, and compliance certifications. The medical-specific sections are what separate successful bids from generic proposals.
Medical courier companies need four types of insurance - general liability, commercial auto, cargo, and inland marine - each covering a different risk category from third-party injury claims to high-value biologics in transit. Coverage requirements vary by contract, and some healthcare facilities specify minimum coverage amounts in their RFPs.
Medical courier contracts pay higher rates than general courier work due to the regulatory requirements, specialized equipment, and liability exposure involved. Per-delivery rates vary by material type, distance, urgency, and region. This guide focuses on winning contracts as a courier company - for individual driver earnings, gig platforms list opportunities.
Healthcare procurement teams score bids on nine criteria, weighting operational evidence over promises. The courier companies that advance to contract negotiation are the ones presenting documented metrics, compliance records, and technology demonstrations - not the ones claiming reliability without proof.
Medical courier contracts reward courier companies that treat operational evidence as their primary sales tool. Procurement teams aren't swayed by promises - they're evaluating documented delivery success rates, compliance readiness, and technology capabilities.
Build your evidence base before you bid. Document everything. Present proof, not claims.
Start your free trial of Spoke Dispatch to see how route optimization, real-time tracking, and electronic proof of delivery strengthen your next contract bid.